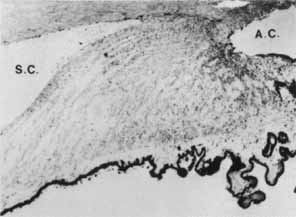
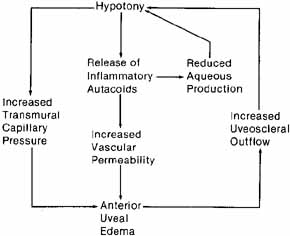
1. Brubaker RF: The physiology of aqueous humour formation. In Drance SM, Neufeld AH (eds): Glaucoma: Applied pharmacology in medical treatment. Orlando: Grune & Stratton, 1984:35–70 2. Bill A, Phillips CI: Uveoscleral drainage of aqueous humor in human eyes. Exp Eye Res 12:275, 1971 3. Pederson JE, Gaasterland DE, MacLellan HM: Uveoscleral aqueous outflow in the rhesus monkey: Importance of uveal reabsorption. Invest Ophthalmol Vis Sci 16:1008, 1977 4. Bill A: The aqueous humor drainage mechanism in the cynomolgus monkey (Macaca irus) with evidence for unconventional routes. Invest Ophthalmol 4:911, 1965 5. Bill A: Conventional and uveo-scleral drainage of aqueous humor in the cynomolgus
monkey (Macaca irus) at normal and high intraocular pressures. Exp Eye Res 5:45, 1966 6. Bill A: Further studies on the influence of the intraocular pressure on aqueous
humor dynamics in cynomolgus monkeys. Invest Ophthalmol 6:364, 1967 7. Phelps CD, Armaly MF: Measurement of episcleral venous pressure. Am J Ophthalmol 85:35, 1978 8. Stepanik J: Die Tonographie und der episklerale Venendruck. Ophthalmologica 133:397, 1957 9. Ramsay RC, Knobloch WH: Ocular perforations following retrobulbar anesthesia for retinal detachment
surgery. Am J Ophthalmol 86:61, 1978 10. Pederson JE, Cantrill HL: Hypotony from occult bridle suture perforation. Arch Ophthalmol 106:581, 1988 11. Savir H: Scleral perforation during cataract surgery. Ann Ophthalmol 15:247, 1983 12. Seelenfreund MH, Freilich DB: Retinal injuries associated with cataract surgery. Am J Ophthalmol 89:654, 1980 13. Bindlish R, Condon GP, Schlosser JD, et al: Efficacy and safety of mitocycin-C in primary trabeculectomy. Ophthalmology 109:1336, 2002 14. Suner IJ, Greenfield DS, Miller MP, et al: Hypotony maculopathy after filtering surgery with mitomycin C. Ophthalmology 104:207, 1997 15. Dellaporta A: Fundus changes in postoperative hypotony. Am J Ophthalmol 40:781, 1955 16. Gass JDM: Hypotony maculopathy. In: Bellows JC (ed): Contemporary ophthalmology, honoring Sir Steward Duke-Elder. Baltimore: Williams and Wilkins, 1972:343–366 17. Kokame GT, deLeon MDL, Tanji T: Serous retinal detachment and cystoid macular edema in hypotony maculopathy. Am J Ophthalmol 131:384, 2001 18. Delgado MF, Daniels S, Pascal S, et al: Hypotony maculopathy: Improvement of visual acuity after 7 years. Am J Ophthalmol 132, 2001 19. Wise J: Treatment of chronic postfiltration hypotony by intrableb injection of
autologous blood. Arch Ophthalmol 111:827, 1993 20. Zaltas MM, Schuman JS: A serious complication of intrableb injection of autologous blood for the
treatment of postfiltration hypotony. Am J Ophthalmol 15:251, 1994 21. Nuyts RM, Greve E, Geijssen C, et al: Treatment of hypotonous maculopathy after trabeculectomy with mitomycin
C. Am J Ophthalmol 118:322, 1994 22. Zacharia PT, Deppermann SR, Schuman JS: Ocular hypotony after trabeculectomy with mitomycin C. Am J Ophthalmol 116:314, 1993 23. Costa VP, Wilson RP, Moster MR, et al: Hypotony maculopathy following the use of topical mitomycin C in glaucoma
filtration surgery. Ophthalmic Surg 24:389, 1993 24. Geijssen HC, Greve EL: Mitomycin C, suture lysis and hypotony. Int Ophthalmol 16:371, 1992 25. Stamper RL, McMenemy MG, Lieberman MF: Hypotonous maculopathy after trabeculectomy with subconjunctival 5-fluorouracil. Am J Ophthalmol 114:544, 1992 26. Cohen SM, Flynn HW Jr, Palmberg PF, et al: Treatment of hypotony maculopathy after trabeculectomy. Ophthalmic Surg Lasers 26:435, 1995 27. Aronson SB, Elliott JH: Ocular inflammation. St. Louis: CV Mosby, 1972:226 28. Pederson JE, Green K: Solute permeability of the normal and prostaglandin-stimulated ciliary
epithelium and the effect of ultrafiltration on active transport. Exp Eye Res 21:569, 1975 29. Weekers R, Delmarcelle Y: Hypotonic oculaire par reduction du debit de 1'humeur aqueuse. Ophthalmologica 125:425, 1953 30. O'Rourke J, Macri FJ: Studies in uveal physiology: II. Clinical studies of the anterior chamber clearance of isotopic tracers. Arch
Ophthalmol 84:415, 1970 31. Toris CB, Pederson JE: Aqueous humor dynamics in experimental iridocyclitis. Invest Ophthalmol Vis Sci 28:477, 1987 32. Brubaker RF, Pederson JE: Ciliochoroidal detachment. Surv Ophthalmol 27:281, 1983 33. Fuchs E: Ablösung der Aderhaut nach Staaroperation. Albrecht Von Graefes Arch Klin Ophthalmol 51:199, 1900 34. O'Brien CS: Detachment of the choroid after cataract extraction. Arch Ophthalmol 14:527, 1935 35. Chylack LT Jr, Bellows AR: Molecular sieving in suprachoroidal fluid formation in man. Invest Ophthalmol Vis Sci 17:420, 1978 36. Capper SA, Leopold IH: Mechanism of serous choroidal detachment. Arch Ophthalmol 55:101, 1956 37. Spaeth EB, DeLong P: Detachment of the choroid: A clinical and histopathologic analysis. Arch Ophthalmol 32:217, 1944 38. Hawkins WR, Schepens CL: Choroidal detachment and retinal surgery. Am J Ophthalmol 62:813, 1966 39. Aaberg TM: Experimental serous and hemorrhagic uveal edema associated with retinal
detachment surgery. Invest Ophthalmol 14:243, 1975 40. Chandler PA, Maumenee AE: A major cause of hypotony. Am J Ophthalmol 52:609, 1961 41. Dellaporta A, Obear MF: Hyposecretion hypotony. Am J Ophthalmol 58:785, 1964 42. Streeten BW, Belkowitz M: Experimental hypotony with Silastic. Arch Ophthalmol 78:503, 1967 43. Pederson JE, Gaasterland DE, MacLellan HM: Experimental ciliochoroidal detachment: Effect on intraocular pressure
and aqueous humor flow. Arch Ophthalmol 97:536, 1979 44. Maruyama Y, Kimura Y, Kishi S, et al: Serous detachment of the ciliary body in Harada disease. Am J Ophthalmol 125:666, 1998 45. Bill A: Movement of albumin and dextran through the sclera. Arch Ophthalmol 74:248, 1965 46. Inomata H, Bill A: Exit sites of uveoscleral flow of aqueous humor in cynomolgus monkey eyes. Exp Eye Res 25:113, 1977 47. Thorpe HE: Diamox in the treatment of non-leaking flat anterior chamber after
cataract extraction and associated with choroidal detachment. Proceedings of the XVII International Congress of Ophthalmology, Vol 3, pp 1855–1864. Toronto, University of Toronto Press, 1955 48. Macri FJ, Brown JG: The constrictive action of acetazolamide on the iris arteries of the cat. Arch Ophthalmol 66:570, 1961 49. Bellows AR, Chylack LT Jr, Hutchinson BT: Choroidal detachment: Clinical manifestation, therapy and mechanism of
formation. Ophthalmology 88:1107, 1981 50. Maumenee AE, Stark WJ: Management of persistent hypotony after planned or inadvertent cyclodialysis. Am J Ophthalmol 71:320, 1971 51. Cadera W, Willis NR: Sodium hyaluronate for postoperative aphakic choroidal detachment. Can J Ophthalmol 17:274, 1982 52. Fisher YL, Turtz Al, Gold Metal: Use of sodium hyaluronate in re-formation and reconstruction of
the persistent flat anterior chamber in the presence of severe hypotony. Ophthalmic Surg 13:819, 1982 53. Shea M, Mednick EB: Ciliary body reattachment in ocular hypotony. Arch Ophthalmol 99:278, 1981 54. Suguro K, Toris CB, Pederson JE: Uveoscleral outflow following cyclodialysis in the monkey eye using a fluorescent
tracer. Invest Ophthalmol Vis Sci 26:810, 1985 55. Bill A: The routes for bulk drainage of aqueous humour in rabbits with and without
cyclodialysis. Doc Ophthalmol 20:157, 1966 56. Goldmann H: Über die Wirkungsweise der Cyclodialyse. Ophthalmologica 121:94, 1951 57. Pederson JE: Ocular hypotony. Trans Ophthalmol Soc UK 105(Part 2):220, 1986 58. Kronfeld PC: The fluid exchange in the successfully cyclodialyzed eye. Trans Am Ophthalmol Soc 52:249, 1954 59. Viikari K, Tuovinen E: On cyclodialysis surgery in the light of follow-up examination. Acta Ophthalmol 35:528, 1957 60. Shaffer RN, Weiss DI: Concerning cyclodialysis and hypotony. Arch Ophthalmol 68:25, 1962 61. Pavlin CJ, Harasiewicz K, Sherar MD, et al: Clinical uses of ultrasound biomicroscopy. Ophthalmology 98:287, 1991 62. Karwatowski WS, Weinreb RN: Imaging of cyclodialysis cleft by ultrasound biomicroscope. Am J Ophthalmol 117:541, 1994 63. Barasch K, Galin MA, Baras I: Postcyclodialysis hypotony. Am J Ophthalmol 68:644, 1969 64. Castier PH, Asseman PH, Razemon L: Evolution d'une hypotonie post-traumatique apres cyclopexie. Bull Soc Ophthalmol Fr 82:261, 1982 65. Kuchle M, Naumann GO: Direct cyclopexy for traumatic cyclodialysis with persisting hypotony. Report
in 29 consecutive patients. Ophthalmology 102:322, 1995 66. Demeler U: Direct cyclopexy after persistent hypotony as a complication after cyclodialysis
and goniotomy. Dev Ophthalmol 13:85, 1987 67. Demeler U: Refixation of the ciliary body after traumatic cyclodialysis. Dev Ophthalmol 14:199, 1987 68. Demeler U: Surgical management of ocular hypotony. Eye 2:77, 1988 69. Naumann GOH, Völcker HE: Direkte Zyklopexie zur Behandlung des persistierenden Hypotonie-Syndroms
infolge traumatisch Zyklodialyse. Klin Monatsbl Augenheilkd 179:266, 1981 70. Zheng Y, Ji X: Reattachment of the detached ciliary body with suturing for treatment of
contusional ocular hypotension. Ophthalmic Surg 22:360, 1991 71. Spiegel D, Katz LF, McNamara JA: Surgical repair of a traumatic cyclodialysis cleft after laser failure. Ophthalmic Surg 21:372, 1990 72. Portney GL, Purcell TW: Surgical repair of cyclodialysis induced hypotony. Ophthalmic Surg 5:30, 1974 73. Joondeph HC: Management of postoperative and post-traumatic cyclodialysis clefts
with argon laser photocoagulation. Ophthalmic Surg 11:186, 1980 74. Harbin TS Jr: Treatment of cyclodialysis clefts with argon laser photocoagulation. Ophthalmology 89:1082, 1982 75. Ormerod LD, Baerveldt G, Sunalp MA, et al: Management of the hypotonous cyclodialysis cleft. Ophthalmology 98:1384, 1991 76. Partamian LG: Treatment of a cyclodialysis cleft with argon laser photocoagulation in
a patient with a shallow anterior chamber. Am J Ophthalmol 99:5, 1985 77. Dobbie JG: A study of the intraocular fluid dynamics in retinal detachment. Arch Ophthalmol 69:159, 1963 78. Burton TC, Arafat NT, Phelps CE: Intraocular pressure in retinal detachment. Int Ophthalmol 1:147, 1979 79. Langham ME, Regan CDJ: Circulatory changes associated with onset of primary retinal detachment. Arch Ophthalmol 81:820, 1969 80. Tulloh GG: The aqueous flow and permeability of the blood-aqueous barrier in
retinal detachment. Trans Ophthalmol Soc UK 92:585, 1972 81. Foulds WS: Experimental detachment of the retina and its effect on the intraocular
fluid dynamics. Mod Probl Ophthalmol 8:51, 1969 82. Pederson JE: Experimental retinal detachment: IV. Aqueous humor dynamics in rhegmatogenous detachments. Arch Ophthalmol 100:1814, 1982 83. Beigelman MD: Acute hypotony in retinal detachment. Arch Ophthalmol 1:463, 1929 84. Pederson JE, Cantrill HL: Experimental retinal detachment: V. Fluid movement through the retinal hole. Arch Ophthalmol 102:136, 1984 85. Ringvold A: Evidence that hypotony in retinal detachment is due to subretinal juxtapapillary
fluid drainage. Acta Ophthalmol 58:652, 1980 86. Barr CC, Lai MY, Lean JS, et al: Postoperative intraocular pressure abnormalities in the Silicone Study: Silicone
Study report 4. Ophthalmology 100:1629, 1993 87. Coleman DJ: Evaluation and management of ciliary body detachment in hypotony. Retina 1:312, 1995 88. O'Connell ST, Majji AB, Humayan MS, et al: The surgical management of hypotony. Ophthalmology 107:318, 2000 89. Duke-Elder S, Jay B: System of Ophthalmology, Vol XI. Diseases of the lens and vitreous; glaucoma and hypotony. St. Louis: CV Mosby, 1969: 724–746 90. Junge J: Ocular changes in dystrophia myotonia, paramyotonia and myotonia congenita. Doc Ophthalmol 21:1, 1966 91. Walker SD, Brubaker RF, Nagataki S: Hypotony and aqueous dynamics in myotonic dystrophy. Invest Ophthalmol Vis Sci 22:744, 1982 92. Raitta C, Karli P: Ocular findings in myotonic dystrophy. Ann Ophthalmol 14:647, 1982 93. Vos TA: 25 years dystrophia myotonic (D.M.). Ophthalmologica 141:37, 1961 94. Radda TM, Bardach H, Riss B: Acute ocular hypotony: A rare complication of temporal arteritis. Ophthalmologica 182:148, 1981 95. Hayreh SS, March W, Phelps CD: Ocular hypotony after retinal vascular occlusion. Trans Ophthalmol Soc U 97:757, 1977 96. Roy FH: Ocular differential diagnosis. Philadelphia: Lea & Febiger, 1993:374–376 97. Blatt N, Regenbogen L: Konsensuelle Hypotonie bei Glaukomoperationen. Klin Monatsbl Augenheilkd 136:761, 1960 98. Völcker HE, Naumann GOH: Morphology of uveal and retinal edemas in acute and persisting hypotony. Mod Probl Ophthalmol 20:34, 1979 99. Pederson JE, MacLellan HM, Gaasterland DE: The rate of reflux fluid movement into the eye from Schlemm's canal
during hypotony in the rhesus monkey. Invest Ophthalmol Vis Sci 17:377, 1978 100. Ambache N, Kavanaugh L, Whiting J: Effect of mechanical stimulation on rabbits' eyes: Release of active
substance in anterior chamber perfusates. J Physiol (Lond) 176:378, 1965 101. Zarbin MA, Michels RG, Green WR: Dissection of epiciliary tissue to treat chronic hypotony after surgery
for retinal detachment with proliferative vitreoretinopathy. Retina 11:208. 1991 102. Boop FE, Lucke K, Laqua H: Resection of the lens capsule by a pars plana approach for the treatment
of ocular hypotony resulting from capsular shrinkage with ciliary body
detachment. Fortschr Ophthalmol 88:802, 1991 103. Daniele S, Schepens CL: Can chronic bulbar hypotony be responsible for uveal effusion? Report
of two cases. Ophthalmic Surg 20:872, 1989 104. Cadera W, Harding PW, Gonder JR, et al: Management of severe hypotony with intravitreal injection of Healon. Can J Ophthalmol 28:236, 1993 105. Morse LS, McCuen BW: The use of silicone oil in uveitis and hypotony. Retina 11:399, 1991 106. Stallman JB, Meyers SM: Repeated fluid-gas exchange for hypotony after vitreoretinal surgery
for proliferative vitreoretinopathy. Am J Ophthalmol 106:147, 1988 107. Krupin T, Weiss A, Becker B, et al: Increased intraocular pressure following topical azide or nitroprusside. Invest Ophthalmol Vis Sci 16:1002, 1977 108. Podos SM: The effect of cation ionophores on intraocular pressure. Invest Ophthalmol Vis Sci 15:851, 1976 109. Macri FJ, Cevario SJ: The induction of aqueous humor formation by the use of ACh + eserine. Invest Ophthalmol Vis Sci 12:910, 1973 110. Bill A, Wahlinder DE: The effects of pilocarpine on the dynamics of aqueous humor in a primate (Macaca irus). Invest Ophthalmol Vis Sci 5:170, 1965 111. Pederson JE, MacLellan HM: Medical therapy for experimental hypotony. Arch Ophthalmol 100:815, 1982 112. Hupsel O, Henkes HE: The treatment of ocular hypotonia with phosphorylcholine chloride: Report
of preliminary experimental and clinical results. In Acta XVII of the International Congress of Ophthalmology, Vol 1. Toronto: University of Toronto Press, 1955: 143–150 113. Boet DJ: Clinical results with phosphorylcholine chloride. Ophthalmologica 132:150, 1956 114. Virno M, Taverniti L, deGregorio F, et al: Increase in aqueous humor production following D1 receptors activation
by means of ibopamine. Internat Ophthalmol 20:141, 1997 |