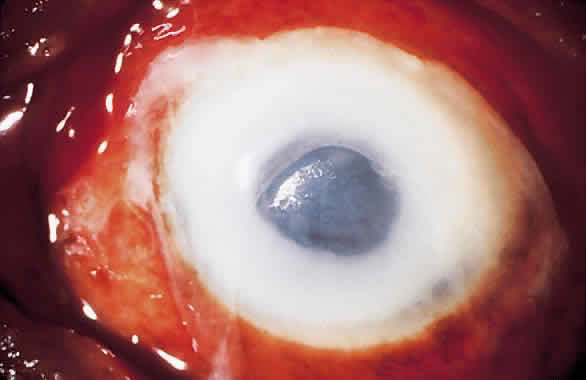
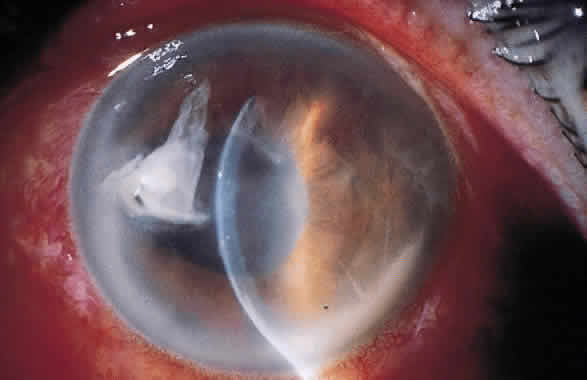
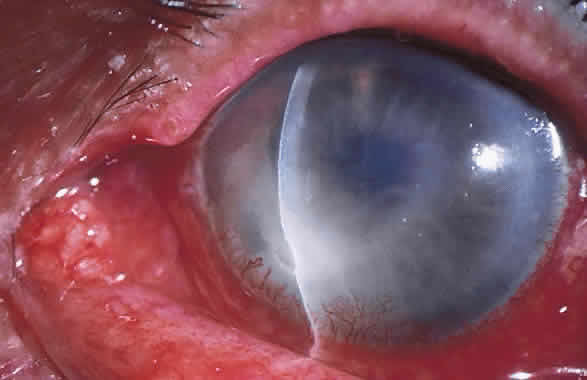
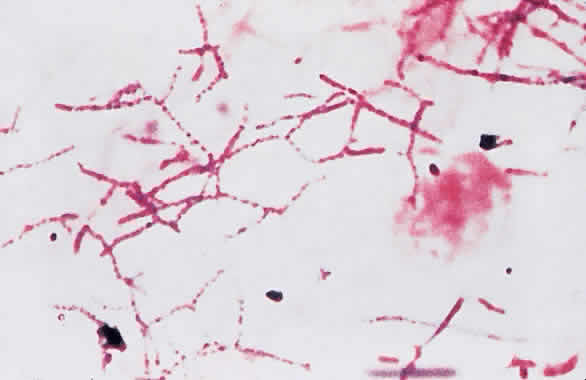
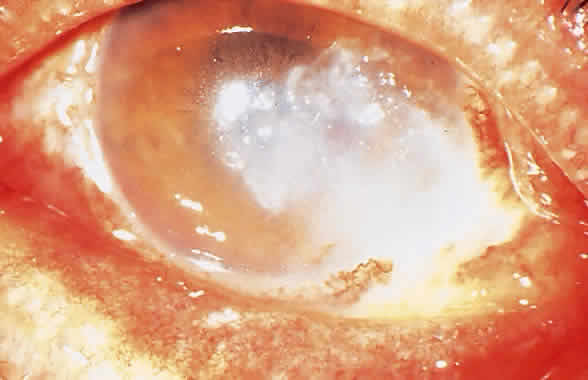
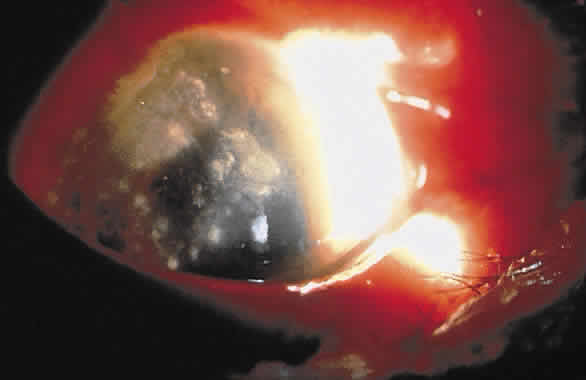
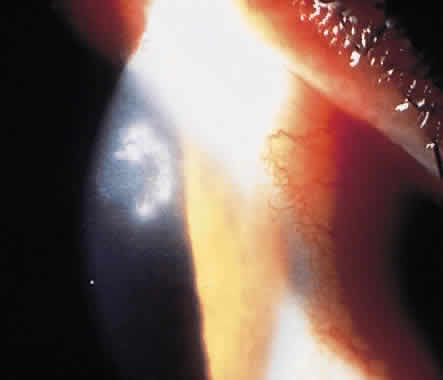
1. Leppla SH: Anthrax toxin edema factor: A bacterial adenylate cyclase that increases
cyclic AMP concentrations in eucaryotic cells. Proc Natl Acad Sci USA 79:3162, 1982 2. Mikesell P, Ivins BE, Ristroph JD et al: Evidence of plasmid-mediated toxin production in Bacillus anthracis. Infect Immun 39:371, 1983 3. O'Day DM, Smith RS, Gregg CR et al: The problem of Bacillus species infection with special emphasis on the virulence of Bacillus cereus. Ophthalmology 88:833, 1981 4. Van Bijsterveld OP, Richards RD: Bacillus infection of the cornea. Arch Ophthalmol 74:91, 1965 5. Donzis PB, Mondino BJ, Weissman BA: Bacillus keratitis associated with contaminated contact lens care systems. Am J Ophthalmol 105:195–197, 1988 6. Tabbara KF, Tarabay N: Bacillus licheniformin corneal ulcer. Am J Ophthalmol 87:717–719, 1979 7. Van Setten GB, Tervo T, Tarkannen A: Akute keratitis und die Kontamination von Kontaktlinsenpflegesystemen mit Bacillus cereus. Klin Monatsbl Augenheilkd 195:28–31, 1989 8. Ormerod LD, Smith RE: Contact lens associated microbial keratitis. Arch Ophthalmol 104:79,1986 9. Donzis PB, Mondino BJ, Weissman BA et al: Microbial contamination of contact lens care systems. Am J Ophthalmol 104:325, 1987 10. McBride ME: Evaluation of microbial flora of the eye during wear of soft contact lenses. Appl Environ Microbiol 37: 233, 1979 11. Debroff BM, Donahue SP, Caputo BJ et al: Clinical characteristics of corneal foreign bodies and their associated
culture results. Cornea 20:128–130, 1994 12. Hollis DG, Weaver RE: Gram Positive Organisms: A Guide to Identification. Centers
for Disease Control, Atlanta, GA, 1981 13. Coyle MB, Hollis DG, Groman NB: Corynebacterium species and other coryneform organisms. In Lennette EH (ed): Manual of
Microbiology, pp 193–204. Washington, DC: American Society for
Microbiology, 1985 14. Lipsky BA, Goldberger AC, Tompkins LS et al: Infections caused by non-diphtheria Corynebacterium. Rev Infect Dis 4:1220, 1982 15. Fedukowicz HB, Stenson S: External Infections of the Eye, pp 2–34. 3rd
ed. Norwalk: Appleton-Century-Crofts, 1985 16. Riley PS, Hollis DG, Utter GB et al: Characterization and identification of 95 diphtheroid (Group JK) cultures
isolated from clinical specimens. J Clin Microbiol 9:418, 1979 17. Funke G, von Graevenitz A, Clarridge III JE et al:Clinical microbiology of coryneform bacteria. Clin Microbiol Rev 10:125–159, 1997 18. Heidemann DG, Dunn SP, Diskin JA et al: Corynebacterium striatus keratitis. Cornea 10:81–82, 1991 19. Funke G, Haase G, Schnitzler N et al: Endophthalmitis due to Microbacterium species: case report and review of
microbacterium infections. Clin Infect Dis 24:713–716, 1997 20. Chandler JW, Milam F: Diphtheria corneal ulcers. Arch Ophthalmol 96:53–57, 1978 21. Rubinfeld RS, Cohen EJ, Arentsen JJ et al: Diphtheroids as ocular pathogens. Am J Ophthalmol 108:251–254, 1989 22. Schuchat A, Swaminathan B, Broome CV: Epidemiology of human listeriosis. Clin Microbiol Rev 4:169–183, 1991 23. Murray EG, Webb RA, Swann MB: A disease of rabbits characterized by a large mononuclear leukocytosis, caused
by a hitherto undescribed bacillus, Bacterium monocytogenes (n.sp.).J Path Bacterial 29:407–439, 1926 24. Anton W: Kritisch-experimentellar Beitrag zur Biologie des Bacterium monocytogenes. Zentralbl Bakteriol Parasitol 131:89–103, 1934 25. Hoeprich PD: Infections caused by Listeria monocytogenes and Erysipelothrix rhusiopathiae.
In Braunwald E, Isselbacher KJ, Petersdarf KG et al (eds): Harrison's
Principles of Internal Medicine, Vol 1. New York: McGraw-Hill, 11:554–556, 1987 26. Skogberg K, Syrjanen J, Jahkola M et al: Clinical presentation and outcome of listeriosis in patients with or without
immunosuppressive therapy. Clin Infect Dis 14:815–821, 1992 27. Sonnenwirth AC, McGee ZA, Davis BD: Other pathogenic microorganisms; L-phase
variants. In Davis BD, Dulbecco R, Einsen HN, Ginsberg HN (eds): Microbiology, pp 797–805. 3rd ed. Philadelphia: Harper & Row, 1980 28. Lohmann CP, Habel VP, Heep M et al: Listeria monocytogenes-induced endogenous endophthalmitis in an otherwise healthy individual: rapid
PCR-diagnosis as the basis for effective treatment. Eur J Ophthalmol 9:53–57, 1999 29. Eiferman RA, Flaherty KT, Rivard AK: Persistent corneal defect caused by Listeria monocytogenes. Am J Ophthalmol 109:97–98, 1990 30. Holbach LM, Bialasiewicz AA, Boltze HJ: Necrotizing ring ulcer of the cornea by exogenous Listeria monocytogenes serotype IV b infection. Am J Ophthalmol 106:105–106, 1988 31. Holland S, Alfonso E, Gelender H et al: Corneal ulcer due to Listeria monocytogenes. Cornea 6:144–146, 1987 32. Zaidman GW, Coudron P, Piros J: Listeria monocytogenes keratitis. Am J Ophthalmol 109:334–339, 1990 33. Pletneva NA, Stiksova VN: Oculoglandular form of listeriosis. Vestn Oftalmol 29:17, 1950 34. Shmeleva VV: Cases of oculoglandular listeriosis. Vestn Oftalmol 32:46, 1953 35. Armstrong D: Listeria monocytogenes. In Mandell GL, Douglas RG, Bennet JE (eds): Principles and Practice of
Infectious Diseases, pp 1587–1593. New York: John Wiley & Sons, 1990 36. Abbott RL, Forster RK, Rebell G: Listeria monocytogenes endophthalmitis with a black hypopyon. Am J Ophthalmol 86:715–719, 1978 37. Algan M, Jonon B, George JL et al: Listeria monocytogenes endophthalmitis in a renal transplant patient receiving cyclosporin. Ophthalmologica 201:23–27, 1990 38. Bagnarello AG, Berlin AJ, Weinstein AJ et al: Listeria monocytogenes endophthalmitis. Arch Ophthalmol 95:1004–1005, 1977 39. Ballen P, Loffredo FR, Paiter B: Listeria endophthalmitis. Arch Ophthalmol 97:101–103, 1979 40. Beuchat L, Hirscel B, Tabatabay C et al: Listeriose intraoculaire. J Fr Ophtalmol 8:797–799, 1985 41. Brasseur G, Retout A, Didier T et al: Endophthalmie à Listeria: une etiologie exceptionelle. Bull Soc Ophtalmol Fr 89:1463–1466, 1989 42. Brian GR, Treplin MCW: Listeria monocytogenes endophthalmitis. Aust N Z J Ophthalmol 16:329–331, 1988 43. Duch ST, Quintana MC, Pujol OG: Listeria monocytogenes endophthalmitis. Acta Ophthalmol 69:108–110, 1991 44. Goodner FK, Okumoto M: Intraocular listeriosis. Am J Ophthalmol 64:682–686, 1967 45. Maloney JM, Nolte FS, Meredith TA: Listeria monocytogenes endophthalmitis, treatment with vitrectomy and trimethoprim/sulfamethoxazole. Emory J Med 4:201–204, 1990 46. Melamed Cattan J, Kwitko S, Barcaro S et al: Listeria monocytogenes endogenous endophthalmitis. Ophthalmology 98: S114, 1991 47. Nigrin J, Tyrrel DLJ, Jackson FL et al: Listeria monocytogenes endophthalmitis in an immunosuppressed host. Can Med Assoc J 116:1378–1382, 1977 48. Snead JW, Stern WH, Whitcher JP et al: Listeria monocytogenes endophthalmitis. Am J Ophthalmol 84:337–340, 1977 49. Eliott D, O'Brien TP, Green WR et al: Elevated intraocular pressure, pigment dispersion and dark hypopyon in
endogenous endophthalmitis from Listeria monocytogenes. Surv Ophthalmol 37:117–124, 1992 50. Gellen BG, Broome CV: Listeriosis. JAMA 261:1313–1320, 1989 51. Hof H, Nichterlein T, Kretschmar M: Management of listeriosis. Clin Microbiol Rev 10:345–357, 1997 52. Brown JM, McNeil MM, Desmond EP: Nocardia, Rhodococcus, Gordona, Actinomadura, Streptomyces, and other actinomycetes of medical importance. In Murray PR, Baron EJ, Pfaller
MA et al (eds): Manual of Clinical Microbiology, pp 370–398. 7th
ed. Washington, DC: American Society for Microbiology, 1999 53. Tsukamura M: Relationship between Mycobacterium and Nocardia. Jap J Microbiol 14:187–195, 1970 54. Lechevalier MP, Horan AC, Lechevalier II: Lipid composition in the classification of Nocardiae and Mycobacteria. J Bacteriol 105:313–318, 1971 55. Bullock JD: Endogenous ocular nocardiosis: a clinical and experimental study. Trans Am Ophthalmol Soc 81:451–531, 1983 56. Benedict WL, Iverson HA: Chronic keratoconjunctivitis associated with Nocardia. Arch Ophthalmol 32:89–92, 1944 57. Climenhaga DB, Tokarewicz AC, Willis NR: Nocardia keratitis. Can J Ophthalmol 19:284–286,1984 58. Donnenfeld ED, Cohen EJ, Barza M et al: Treatment of Nocardia keratitis with topical trimethoprim-sulfamethoxazole. Am J Ophthalmol 99:601–602, 1985 59. Gingrich WD: Keratomycosis. JAMA 179:602–608, 1962 60. Hirst LW, Harrison KG, Merz WG et al: Nocardia asteroides keratitis. Br J Ophthalmol 63:449–454, 1979 61. Hirst LW, Merz WG, Green WR: Nocardia asteroides corneal ulcer. Am J Ophthalmol 94:123–124, 1982 62. Newmarck E, Polack FM, Ellison AC: Report of a case of Nocardia asteroides keratitis. Am J Ophthalmol 72:813–815, 1971 63. Penikett EJK, Rees DL: Nocardia asteroides infection. Am J Ophthalmol 53:1006–1008, 1962 64. Ralph RA, Lemp MA, Liss G: Nocardia asteroides keratitis: A case report. Br J Ophthalmol 60:104–106, 1976 65. Schardt WM, Unsworth AC, Hayes CV: Corneal ulcer due to Nocardia asteroides. Am J Ophthalmol 42:303–305, 1956 66. Sigtenhorst ML, Gingrich WD: Bacteriologic studies of keratitis. South Med J 50:346–350, 1975 67. Zimmerman LE: Mycotic keratitis. Lab Invest 11:1151–1160, 1962 68. Zimmerman LE: Keratomycosis. Surv Ophthalmol 8:1–25, 1963 69. McLean JM: Oculomycosis. Trans Am Acad Ophthalmol Otolaryngol 67:149–163, 1963 70. Archibald RG: Primary nocardiosis of the lacrimal gland. Lancet 2:847, 1918 71. McCarthy JL, Kennedy RJ, Hazard JB: Experimental ocular infection with Nocardia asteroides in the normal rabbit. Arch Ophthalmol 62:425–433, 1959 72. Rogers SJ, Johnson BL: Endogenous Nocardia endophthalmitis: report of a case in a patient treated for lymphocytic
lymphoma. Ann Ophthalmol 9:1123–1131, 1977 73. Sher NA, Hill CW, Eifrig DE: Bilateral intraocular Nocardia asteroides infection. Arch Ophthalmol 95:1415–1418, 1977 74. Burpee JC, Starke WR: Bilateral metastatic intraocular nocardiosis. Arch Ophthalmol 86:666–669, 1971 75. Davidson S, Foerster HC: Intraocular nocardial abscess, endogenous. Trans Am Acad Ophthalmol Otolaryngol 71:847–850, 1967 76. Jampol IM, Strauch BS, Albert DM: Intraocular nocardiosis. Am J Ophthalmol 76:568–573, 1973 77. Meyer SL, Font RL, Shaver RP: Intraocular nocardiosis: report of three cases. Arch Ophthalmol 83:536–541, 1970 78. Sher NA, Hill CW, Eifrig DE: Bilateral intraocular Nocardia asteroides infection. Arch Ophthalmol 95:1415–1418, 1977 79. Lissner GS, O'Grady R, Chromokos E: Endogenous intraocular Nocardia asteroides in Hodgkin's disease. Am J Ophthalmol 86:388–394, 1978 80. Panijayanond P, Olsson CA, Spivack ML et al: Intraocular nocardiosis in a renal transplant patient. Arch Surg 104:845–847, 1972 81. Boiron P, Provost F: In vitro susceptibility testing of Nocardia spp. and its taxonomic implication.J Antimicrob Chemother 22:623–629, 1988 82. Dewsnup DH, Wright DN: In vitro susceptibility of Nocardia asteroides to 25 antimicrobial agents. Antimicrob Agents Chemother 25:165–167, 1984 83. Kitzias MD, Gutmann L, Acan JF: In vitro susceptibility of Nocardia asteroides to 21 β-lactam antibiotics, in combination with three β-lactamase
inhibitors and its relationship to the β-lactamase
content. J Antimicrob Chemother 15:23–30, 1985 84. Lerner PI, Baum GL: Antimicrobial susceptibility of Nocardia species. Antimicrob Agents Chemother 4:85–93, 1973 85. Wallace RJ, Septimus EJ, Musher DM et al: Disk diffusion susceptibility testing of Nocardia species. J Infect Dis 135:568–576, 1977 |